Beware the Switch Flippers
microRNA target sites on Pfizer and Moderna vaccines are very concerning
“One does not become enlightened by imagining figures of light, but by making the darkness conscious.” - Carl Jung
This article is an exercise in imagination through which we will, I believe, establish a compelling conceptual model for the workings of the mRNA vaccines. I want to make it clear up front that I am not an “expert” in the biological sciences. Therefore, there may very well be important details that I miss in my analysis. Even so, it is, frankly, difficult for me to imagine that the model I will be proposing is fatally flawed in some way. I feel strongly that this model is both valid and incredibly important for all of us to think deeply about.
Let us imagine that the following statements are true:
The mRNA vaccines for covid-19 readily distribute systemically to virtually all major organs, in particular, the heart. Although, shockingly, this has not yet been studied in detail, there is supporting evidence for wide-scale biodistribution from a study performed by researchers in Japan1. Also, as Seneff et al. point out, large quantities of circulating exosomes containing vaccine mRNA have been found following vaccination2.
Uncontrolled expression of spike protein in heart tissues almost invariably leads to severe acute myocarditis. As of this writing, vaccine-induced myocarditis occurs at an apparently low rate in the population (~ 12.6 cases per million doses)3. However, in the mouse model, intravenous vaccination with Pfizer BNT162b2 produced severe myocarditis4, suggesting, in principle at least, that the mRNA vaccines are primed to produce severe inflammation in the heart leading to myocarditis.
Vaccine manufacturers were aware of both 1 and 2 above during the design phase and they took active measures to minimize the risk of acute myocarditis following vaccination. After all, such an obvious clinical manifestation which carries such a high risk of adverse outcomes and death would quickly put an end to the campaign to vaccinate the entire world.
If you were the vaccine manufacturer how would you approach this problem? I suppose most of us are decent enough to admit to ourselves that the product is just too dangerous to release. But, remember, you are an evil person intent on pushing your product upon the whole world. All you need to do is find a mechanism to incorporate into your vaccine that will actively suppress myocarditis.
Light Bulb!
I’d like to propose to you, Mr. (Mrs.) Evil Vaccinator, that one very effective way of accomplishing your objective of minimizing myocarditis is to use microRNA as a switch that will turn off expression of the spike protein before heart inflammation becomes too extreme. Do you want to hear more?
Ok, then, let’s start with some basics.
The Master Switch
microRNAs (often abbreviated as miRNA or miR) are just tiny strands of non-coding RNA, usually around 21 to 23 nucleotides, but they are incredibly powerful master regulators of all kinds of physiological processes in the body. Just think of microRNA as a simple switch:
The mechanism by which microRNAs accomplish their remarkable switching ability is by binding with transcribed mRNAs floating around in the cytoplasm. Once a particular microRNA binds to a target site on a mRNA strand, expression of the strand becomes blocked and the mRNA just naturally decays. In this manner, microRNA exerts direct control over protein expression. Protein expression can be up-regulated by down-regulating its corresponding microRNA, and vice-versa. This is how microRNA acts as a switch. It can literally turn genes on and off. In computer terminology, if DNA/RNA represents the assembly code of life, then microRNA would be the processor registers. Understanding those registers and how they impact the behavior of the processor (human body) yields incredible power to influence the system in a desired manner.
Considering the fact that we only discovered the high-level function of microRNA around the year 2000, there is still a great deal that we do not yet understand. So far, we’ve identified over 2000 unique microRNAs in the human body, but we’ve only studied the switching functions of a small fraction of them. Moreover, each microRNA can potentially target the expression of hundreds of target genes5.
One thing we do know is that microRNAs play a profound role in the development and maintenance of the healthy human being. Certain microRNAs tend to be associated with different tissues and organs where they regulate physiological processes. Naturally, problems can be expected to arise when the expression of microRNAs becomes dysregulated. Just about any disease you can imagine can be traced to disturbances in microRNA regulation including cancer, heart disease, kidney disease and neurodegeneration. It has also been found that microRNAs can be transmitted between cells via exosomes, which provides a mechanism by which disease or stress conditions can be communicated systemically6.
Taking the Reins
We humans are insatiable when it comes to the degree of control we exert over everything around us (and in us). So, naturally, after discovering the pivotal role of microRNA in biology, our attention immediately turned towards finding ways to control the switching of microRNA ourselves. Depending on the direction of the dysregulation that needs to be corrected by therapeutic intervention, we can make use of microRNA enhancers or suppressors. For enhancement, typically a microRNA mimic or precursor is administered to the target tissue/organ. On the other hand, suppression can be achieved by administering an inhibitor or sponge7.
The mRNA sponge is of the most interest to us here. It’s a very simple idea. If you want to sop up a particular microRNA then one really good way to do that is to administer a strand of mRNA (usually non-coding) that has one or more target sites for the microRNA of interest. An example application under active investigation would be the use of a sponge to decrease the levels of microRNAs associated with cancer progression8. Of particular note, it has been found that mRNA sponges containing the N1-methylpseudouridine substitution outperform sponges containing the uridine base owing to the increased hydrogen bonding provided by the former9. If you weren't aware, it just so happens that both the Pfizer and Moderna mRNA vaccines utilize the N1-methylpseudouridine substitution10.
The Vaccines are Sponges!
In one of life’s strange coincidences, in the process of researching this topic I came across a stellar paper by Dr. Kevin Harrod11. If you haven’t read any of my other articles, I have written about him quite a bit in connection to SARS-CoV research performed at Lovelace Respiratory Research Institute in Albuquerque, NM. So, needless to say, I was quite surprised to come across his paper which essentially laid out the template for how to analyze microRNA binding to target mRNAs.
The paper was published in August 2020 and it was focused on searching for microRNAs which might have a high probability of binding to the SARS-CoV-2 genome. The idea, which I find fascinating, is that most of the pathological effects of SARS-CoV-2 infection might arise due to the viral RNA acting as a sponge. Wow.
Dr. Harrod provided me the tool I was looking for to perform the exact same analysis on the vaccine RNA sequences - the RNA22 v2 microRNA target discovery tool. This algorithm examines a mRNA sequence for potential binding sites for a particular microRNA and ranks them according to their statistical P-values.
I obtained the putative sequences for the spike protein-encoding mRNA in the Pfizer BNT-162b2 and Moderna mRNA-1273 from Github and the spike protein sequence for the natural Wuhan strain from GenBank. Next, I grabbed the sequences of the following microRNAs from miRBase: miR-1, miR-21, miR-29a/b/c, miR-33a, miR-122, miR-129, miR-132, miR-133a, miR-133b, miR-148b, miR-185, miR-192, miR-199a, miR-199b, miR-208a, miR-208b, miR-214, miR-223 and miR-425. I chose these particular microRNAs because they are all involved in the physiology of the heart, which was my primary focus in this analysis. I used the same strict settings as Harrod in order to reduce false matches (92% specificity, 22% sensitivity). Moreover, I only considered matches that gave a P-value less than or equal to 0.04.
Here’s a table showing the results:
1213141516171819202122232425262728293031323334353637383940414243444546474849505152535455565758
Well, right off the bat you can see something very strange in the table above. There are no matches for the wild-type Wuhan spike protein, but several for both the Pfizer and Moderna spikes. The only reasonable conclusion would be that those target sites were intentionally included in the vaccine sequences!
The big question is why? Well, as you can see in the column on the right side of the table, I’ve taken my best guess as to the putative purpose for each microRNA. My proposition is that these targets have been added mostly to manage and suppress acute myocarditis. By acting as a sponge, the vaccine mRNA can soak up harmful microRNAs that arise due to the inflammatory response in the heart. But, more importantly, the expression of the spike can in effect be regulated based on the feedback signal arising from mycarditis-related microRNAs. This is an incredibly important function that we need to discuss in more detail.
Myocarditis on cruise-control
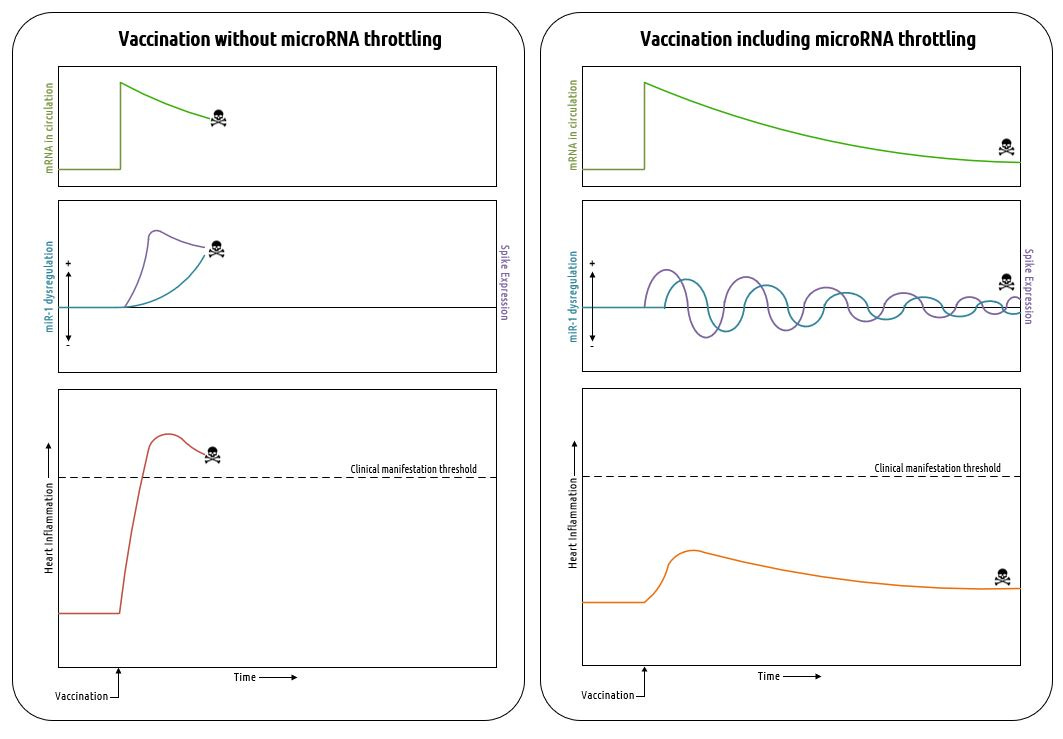
I’m going to show you a hypothetical graph that may explain everything we have been observing in the vaccinated population over the last couple of years.
In the left panel, you can see a possible outcome of vaccination with mRNA that does not include microRNA target sites. Without these sites, particularly the ones which bind to myocarditis-related microRNAs (e.g. miR-1, miR-21), there is no feedback mechanism available to regulate spike protein expression in the heart. This can lead to uncontrolled spike expression followed by uncontrolled inflammation of the heart. The prognosis for severe acute myocarditis is not good, not to mention it is a highly visible condition that would interfere with the plan to vaccinate the world.
The possible effects of including microRNA target sites are shown in the right panel. Here, we see an underdamped, ringing response in the levels of miR-1 (I’ve used this particular microRNA as an example, but it’s important to note that a few others are involved with heart inflammation) along with spike expression in the heart. The expression of spike protein leads the miR-1 response because as the amount of spike protein increases, inflammation increases and produces an increase in miR-1. The miR-1 binds with the target site on the vaccine mRNA and suppresses its expression. As miR-1 is soaked up by the mRNA sponge, its levels decrease until new mRNA from circulation enters the heart and turns on spike expression again to repeat the cycle. The amplitudes of the oscillations in miR-1 and spike protein attenuate over time as the amount of mRNA in circulation decays. The overall result of this process is to produce an attenuated level of inflammation in the heart, otherwise known as chronic myocarditis.
Chronic myocarditis is no joke. Extended inflammatory processes in the heart promote cardiac fibrosis and dilated cardiomyopathy. Fibrosis, in particular, can lead to the development of arrhythmias and sudden cardiac death59. Also, as you may have noted in the table I presented previously, some of the microRNAs actually promote fibrosis when soaked up by a sponge. You have to wonder if this was intentional.
I have to stress here that this is purely a thought-experiment at this point. My thinking has been guided by what we have been observing in the vaccinated population along with the discovery that important microRNA binding sites are present in the vaccines but strangely absent in the wild-type spike protein sequence.
Still, though, it seems quite reasonable and compelling to me. If this is in fact what is truly happening, it would appear that the real curve that was flattened during the covid pandemic was the one for vaccine-induced myocarditis. Acute deaths from the vaccine were suppressed, but the long-term risk from chronic myocarditis was the cost.
The Cancer Connection
I have been primarily focused on the heart in this discussion, but we should probably take a moment to address the effect of microRNA sponges on cancer progression. Most of the microRNAs in the table above actually promote the proliferation of cancer cells when soaked up by a sponge, and most of them are present in a variety of different tissues/organs. It’s important to consider that the vaccine mRNA is delivered all over the body. If it is actively soaking up microRNAs that suppress tumor growth then we should not at all be surprised to see an uptick in cancers.
Summary
My biggest goal here was to hopefully spark discussion of what I consider to be an extremely important topic. We desperately need to coalesce around a comprehensive and cohesive conceptual model that adequately explains the wide range of phenomena that we are observing now as it relates to the mRNA vaccines. Most importantly, we need truth in this world. I don’t know about you, but I’ve had more than ten lifetimes worth of bullshit. I’m about over it.
Please reach out if you feel that I have anything obviously wrong or if you have suggestions about how to improve the presentation.
Thanks for reading. God bless and take care.
Seneff, Stephanie, et al. "Innate immune suppression by SARS-CoV-2 mRNA vaccinations: The role of G-quadruplexes, exosomes, and MicroRNAs." Food and Chemical Toxicology 164 (2022): 113008.
Bozkurt, Biykem, Ishan Kamat, and Peter J. Hotez. "Myocarditis with COVID-19 mRNA vaccines." Circulation 144.6 (2021): 471-484.
Li, Can, et al. "Intravenous injection of coronavirus disease 2019 (COVID-19) mRNA vaccine can induce acute myopericarditis in mouse model." Clinical Infectious Diseases 74.11 (2022): 1933-1950.
Hammond SM. An overview of microRNAs. Adv Drug Deliv Rev. 2015 Jun 29;87:3-14. doi: 10.1016/j.addr.2015.05.001. Epub 2015 May 12. PMID: 25979468; PMCID: PMC4504744.
O'Brien, Jacob, et al. "Overview of microRNA biogenesis, mechanisms of actions, and circulation." Frontiers in endocrinology 9 (2018): 402.
Hum, Christine, et al. "MicroRNA mimics or inhibitors as antiviral therapeutic approaches against COVID-19." Drugs 81.5 (2021): 517-531.
Tay, Felix Chang, et al. "Using artificial microRNA sponges to achieve microRNA loss-of-function in cancer cells." Advanced drug delivery reviews 81 (2015): 117-127.
Parr, Callum JC, et al. "N 1-Methylpseudouridine substitution enhances the performance of synthetic mRNA switches in cells." Nucleic acids research 48.6 (2020): e35-e35.
Morais P, Adachi H, Yu YT. “The Critical Contribution of Pseudouridine to mRNA COVID-19 Vaccines.” Front Cell Dev Biol. 2021 Nov 4;9:789427.
Bartoszewski, Rafal, et al. "SARS-CoV-2 may regulate cellular responses through depletion of specific host miRNAs." American Journal of Physiology-Lung Cellular and Molecular Physiology 319.3 (2020): L444-L455.
Li, Wei, et al. "MiR-1/133 attenuates cardiomyocyte apoptosis and electrical remodeling in mice with viral myocarditis." Cardiology Journal 27.3 (2020): 285-294.
Safa, Amin, et al. "miR-1: A comprehensive review of its role in normal development and diverse disorders." Biomedicine & Pharmacotherapy 132 (2020): 110903.
Chang, Chia-Yu, et al. "Roles of microRNA-1 in hypoxia-induced apoptotic insults to neuronal cells." Archives of toxicology 90 (2016): 191-202.
Wang, Yu, et al. "Astragalus Root dry extract restores connexin43 expression by targeting miR-1 in viral myocarditis." Phytomedicine 46 (2018): 32-38.
Surina, Surina, et al. "miR-21 in human cardiomyopathies." Frontiers in Cardiovascular Medicine 8 (2021): 767064.
Shioya, Mika, et al. "Aberrant microRNA expression in the brains of neurodegenerative diseases: miR‐29a decreased in Alzheimer disease brains targets neurone navigator 3." Neuropathology and applied neurobiology 36.4 (2010): 320-330.
Yang, Zeran, et al. "MiR-29a modulates the angiogenic properties of human endothelial cells." Biochemical and biophysical research communications 434.1 (2013): 143-149.
Xuan, Ji, et al. "MiR-29a and miR-652 attenuate liver fibrosis by inhibiting the differentiation of CD4+ T cells." Cell Structure and Function 42.2 (2017): 95-103.
Horita, Masahiro, Colin Farquharson, and Louise A. Stephen. "The role of miR‐29 family in disease." Journal of cellular biochemistry 122.7 (2021): 696-715.
Vitry, Geraldine, et al. "Mir-33: miR-acles in cardiac fibrosis?." Age 18: 19.
Zhou, Jianda, et al. "miR-33a functions as a tumor suppressor in melanoma by targeting HIF-1α." Cancer biology & therapy 16.6 (2015): 846-855.
Zhang, Chuankai, et al. "MiR-33a suppresses breast cancer cell proliferation and metastasis by targeting ADAM9 and ROS1." Protein & cell 6.12 (2015): 881-889.
Liu, Ying, et al. "Roles of microRNA-122 in cardiovascular fibrosis and related diseases." Cardiovascular Toxicology 20 (2020): 463-473.
Wei, Qi, et al. "Long noncoding RNA NEAT1 promotes myocardiocyte apoptosis and suppresses proliferation through regulation of miR-129-5p." Journal of Cardiovascular Pharmacology 74.6 (2019): 535-541.
Wu, Junjie, et al. "miR-129 regulates cell proliferation by downregulating Cdk6 expression." Cell cycle 9.9 (2010): 1809-1818.
Wang, Feng, et al. "Inhibition of miR-129 improves neuronal pyroptosis and cognitive impairment through IGF-1/GSK3β signaling pathway: an in vitro and in vivo study." Journal of Molecular Neuroscience (2021): 1-11.
Foinquinos, Ariana, et al. "Preclinical development of a miR-132 inhibitor for heart failure treatment." Nature communications 11.1 (2020): 633.
Majer, Anna, et al. "Early mechanisms of pathobiology are revealed by transcriptional temporal dynamics in hippocampal CA1 neurons of prion infected mice." PLoS pathogens 8.11 (2012): e1003002.
Huang, Jiansheng, et al. "MicroRNA‑132‑3p regulates cell proliferation, apoptosis, migration and invasion of liver cancer by targeting Sox4." Oncology letters 19.4 (2020): 3173-3180.
Hua, Yu-Ting, et al. "Emerging roles of MiR-133a in human cancers." Journal of Cancer 12.1 (2021): 198.
Zhu, Yi-Fan, et al. "miR-133a-3p attenuates cardiomyocyte hypertrophy through inhibiting pyroptosis activation by targeting IKKε." Acta Histochemica 123.1 (2021): 151653.
Li, Ning, Heng Zhou, and Qizhu Tang. "miR-133: a suppressor of cardiac remodeling?." Frontiers in pharmacology 9 (2018): 903.
Wu, Zheng-sheng, et al. "Loss of miR-133a expression associated with poor survival of breast cancer and restoration of miR-133a expression inhibited breast cancer cell growth and invasion." BMC cancer 12.1 (2012): 1-10.
Yang, Qin, Qiuling Zhao, and Yanliang Yin. "miR‑133b is a potential diagnostic biomarker for Alzheimer's disease and has a neuroprotective role." Experimental and Therapeutic Medicine 18.4 (2019): 2711-2718.
Xiang, Kai-Min, and Xiao-Rong Li. "MiR-133b acts as a tumor suppressor and negatively regulates TBPL1 in colorectal cancer cells." Asian Pacific Journal of Cancer Prevention 15.8 (2014): 3767-3772.
Zhang, Song Lin, et al. "Effect of microRNA-133b on myocardial fibrosis." Zhongguo yi xue ke xue Yuan xue bao. Acta Academiae Medicinae Sinicae 41.5 (2019): 589-594.
Sun, Hualing, et al. "miR-148-3p: mdfic Pathway Improves the Efficacy of Therapeutic Cardiac Reprogramming." Circulation 146.Suppl_1 (2022): A12047-A12047.
Friedrich, Michael, et al. "The role of the miR‐148/‐152 family in physiology and disease." European journal of immunology 47.12 (2017): 2026-2038.
Lin, Ruizhu, et al. "MiR-185-5p regulates the development of myocardial fibrosis." Journal of Molecular and Cellular Cardiology 165 (2022): 130-140.
Serafini, Gianluca, et al. "The involvement of microRNAs in major depression, suicidal behavior, and related disorders: a focus on miR-185 and miR-491-3p." Cellular and molecular neurobiology 34 (2014): 17-30.
Li, Shuai, et al. "MiR-185 acts as a tumor suppressor by targeting AKT1 in non-small cell lung cancer cells." International journal of clinical and experimental pathology 8.9 (2015): 11854.
Ren, Fu-jia, et al. "Emerging role of MiR-192-5p in human diseases." Frontiers in Pharmacology 12 (2021): 614068.
Flammang, Isabelle, et al. "Tumor-suppressive miR-192-5p has prognostic value in pancreatic ductal adenocarcinoma." Cancers 12.6 (2020): 1693.
Zeng, Ni, et al. "Diverging targets mediate the pathological role of miR-199a-5p and miR-199a-3p by promoting cardiac hypertrophy and fibrosis." Molecular Therapy-Nucleic Acids 26 (2021): 1035-1050.
He, Jun, et al. "Roles and mechanism of miR-199a and miR-125b in tumor angiogenesis." PloS one 8.2 (2013): e56647.
Li, Zhenhua, et al. "miR-199-sponge transgenic mice develop physiological cardiac hypertrophy." Cardiovascular research 110.2 (2016): 258-267.
Duygu, Burcu, et al. "miR-199b-5p is a regulator of left ventricular remodeling following myocardial infarction." Non-coding RNA research 2.1 (2017): 18-26.
Fang, Cheng, et al. "Down-regulation of miR-199b-5p is correlated with poor prognosis for breast cancer patients." Biomedicine & Pharmacotherapy 84 (2016): 1189-1193.
Montgomery, Rusty L., et al. "Therapeutic inhibition of miR-208a improves cardiac function and survival during heart failure." Circulation 124.14 (2011): 1537-1547.
Zou, Yutian, et al. "circRAD18 sponges miR-208a/3164 to promote triple-negative breast cancer progression through regulating IGF1 and FGF2 expression." Carcinogenesis 40.12 (2019): 1469-1479.
Soci, Ursula Paula Renó, et al. "Epigenetic control of exercise training-induced cardiac hypertrophy by miR-208." Clinical Science 130.22 (2016): 2005-2015.
Yang, Kun, et al. "The deficiency of miR-214-3p exacerbates cardiac fibrosis via miR-214-3p/NLRC5 axis." Clinical Science 133.17 (2019): 1845-1856.
Dettori, Daniela, et al. "Therapeutic silencing of miR-214 inhibits tumor progression in multiple mouse models." Molecular Therapy 26.8 (2018): 2008-2018.
Zhang, Miaoyu, et al. "Up-regulating microRNA-214-3p relieves hypoxic–ischemic brain damage through inhibiting TXNIP expression." Molecular and Cellular Biochemistry (2022): 1-12.
van Balkom, Bas WM, et al. "Endothelial cells require miR-214 to secrete exosomes that suppress senescence and induce angiogenesis in human and mouse endothelial cells." Blood, The Journal of the American Society of Hematology 121.19 (2013): 3997-4006.
Zhang, Meng-Wan, et al. "MiR-223-3p in cardiovascular diseases: a biomarker and potential therapeutic target." Frontiers in Cardiovascular Medicine 7 (2021): 610561.
Wang, Lu, et al. "miR-425 reduction causes aberrant proliferation and collagen synthesis through modulating TGF-β/Smad signaling in acute respiratory distress syndrome." International Journal of Clinical and Experimental Pathology 12.7 (2019): 2604.
Ali-Ahmed F, Dalgaard F, Al-Khatib SM. Sudden cardiac death in patients with myocarditis: Evaluation, risk stratification, and management. Am Heart J. 2020 Feb;220:29-40.